EDI Technology
The advanced EDI technology is typically implemented downstream Reverse Osmosis (RO) equipment and mainly used to produce High Purity and Ultra Pure Water (UPW). The environmental friendly operating combined RO-EDI systems, as the successor for the more traditional Ion Exchange resin installations, is nowadays the standard for the production of demineralized water. EDI equipment can operate continuously and produces a consistent quality of pure water. The systems have a very limited footprint and frequent maintenance is not applicable. Electricity is all that is needed for the electrochemical process operation.

We sell products from




The EDI technology to produce ultrapure water
Companies constantly focus on the reduction of operating costs, to improve efficiency and eliminate the use of hazardous chemicals in the workplace. Such goals embraced the increasing implementation of the EDI technology to produce high purity and Ultra Pure Water.
Even though in some sectors EDI is still considered as a new technology, in reality, many thousands of systems are operating successfully in different environments for 25 years. In these periphrases the EDI history, the basic principles and features are further explained.
EDI History
EDI is first described in a scientific publication in 1955 as a method to remove trace radioactive materials from water (Walters, et al.) and until the first commercial system in early 1987 many studies and patents have been developed. Since 1987 until today several commercial EDI manufacturers entered the market. Since then the interest in EDI technology was fostered due to the rapidly emerging needs of tomorrow and the implementation of Corporate Social Responsibility.
Questions about our products?
EDI Applications
Electrodeionization is the technology of choice for projects that require pure product water and stringent wastewater discharge requirements. Among several applications is the production of demineralized process water for the power and petrochemical industry including (high pressure) boiler feed water and pure water injection in gas turbines. Other typical EDI pure water processes are for the semi-conductor industry and optical-glass manufacturing. Since decades the EDI technology is the standard technology as the final step for the production of Purified Water according to the guidelines of the United States (USP) and European Pharmacopoeia (EP). During operation, EDI modules are at least considered bacteriostatic and additionally these can be frequently hot water or chemical sanitized.
Typical applications of EDI are:
- Demineralized feed water for (supercritical high pressure) boilers including Concentrated Solar Plants.
- Pure water injection in turbines for NOx reduction.
- Microelectronics / Semiconductor rinse water.
- Purified and Highly Purified Water for the pharmaceutical industry.
- Demineralized water for the (petro)chemical industry.
- Hospital applications including Haemodialysis.
- University and laboratory.


How EDI is built and operates
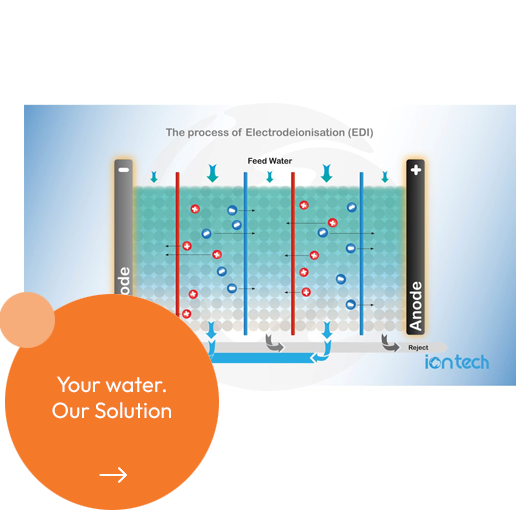
Electrodeionization (EDI) is a process that removes ions such as salts, acids and bases. Weakly ionized materials like dissolved silica, carbon dioxide, boron and some organics are removed as well. The bulk of the ions is removed in the upper part of the module (working bed) and transported via the conductive surface of the ion exchange (IX) resin beads towards the IX membranes. The applied DC current is the driving force for the ion transport to and migration through the IX membranes into the concentrate compartments.
In the lower part of the module, the so-called “polishing bed”, the remaining ions and especially the weakly charged ions are removed by the “ion exchange” phenomena. The ion exchange process is driven by continuous water splitting (electrical regeneration). To further explain the process in detail let us build a basic EDI module. First, a container is filled with water containing salts and additionally on the two ends two electrodes connected to a DC power supply are introduced.
Under the influence of the applied electrical potential (DC Voltage) the negative anions and the positive cations move towards the anode and cathode respectively. To be able to separate the anions (negative charge) and cations (positive charge), a number of ion-selective IX membranes are introduced in the container as well, creating alternately “High Purity product” and “Concentrate” compartments. As a result of the transport of anions in one direction and of cations in the opposite direction, ions can pass one membrane and are then repelled by the next membrane. In this way, all ions are trapped in the concentrate compartments. In an EDI module a part of the feed flow, approximately 5% of this feed flow, is used to continuous wash out the ions from the concentrate compartments. This concentrate outlet, also called reject stream, can be recirculated upstream the RO system to be recovered.
Water splitting
As most of the ions are already removed in the upper part of the product compartments, the electrical resistance between membranes and resin beads increases significantly in the lower part. As a result, also the local voltage potential increases and at a certain voltage difference, water splitting occurs. Water (H2O) splits into hydrogen (H+) and hydroxide (OH–) ions. The H+ and OH– ions replace trace anions of elements like sodium and chloride and weakly charged ions like silica, boron and CO2 on the resin beads so these can escape into the concentrate compartments. More than 80% of the power is used for water splitting and possible excess of H+ and OH– ions also move into the concentrate compartments and form H2O again.


This basic repeating element of the EDI, called a “cell-pair”, is illustrated above. The “stack” of cell-pairs is positioned between the two electrodes. Under the influence of the applied DC voltage potential, ions are transported across the membranes from the product compartments into the concentrate compartments. Thus, as water moves through the product compartments, these become free of ions. This stream is the pure water product stream. In the concentrate stream, electrical neutrality is maintained. Transported ions from the two directions neutralize one another’s charge.
In an EDI device, the compartments are filled with electrically active media such as ion exchange resin. The IX resin enhances the transport of ions and can also participate as a substrate for electrochemical reactions, such as splitting of water into hydrogen (H+) and hydroxyl (OH–) ions.
Different media configurations are possible and used by different manufacturers, such as intimately mixed anion and cation exchange resins (mixed bed or MB). Or separate sections of ion-exchange resin, each section substantially comprised of resins of the same polarity: e.g., either anion or cation resin (layered bed or LB and single bed or SB). The electrical water dissociation produces the continuous regeneration of resins by hydrogen and hydroxide ions. This dissociation preferentially occurs at bipolar interfaces in the ion-depleting compartment where localized conditions of low solute concentrations are most likely to occur (Simons). Regenerating the resins to their H+ and OH– forms allow EDI devices to remove weakly ionized compounds such as carbonic and silicic acids and to remove weakly ionized organic compounds.
Beneficial EDI characteristics
The electrodeionization technology distinguishes itself as a continuous ion exchange technique for the production of (ultra) pure water without the need for chemical regeneration and wastes neutralization steps. During the process, the polishing part of the resin is continuously regenerated. In comparison with the more traditional ion exchange systems, this reduces facility costs such as waste neutralization equipment and hazardous fumes ventilation. The lack of chemical usage, handling and storage help to improve health and safety, as well as corrosion prevention in facilities and equipment. EDI modules can be fed with feed water conductivity equivalent up to 40 µS/cm and water hardness up to 1 ppm, allowing a wider range of applications. Electro deionization systems can achieve up to 99,9 % salt removal, reduce the levels of individual ionic species to parts-per-billion or even parts-per-trillion levels, and produce high-purity water with a resistivity of 10 to 18,2 MegOhm.cm (0,1 to 0,055 microS/cm conductivity). Since the EDI concentrate (or reject) stream contains the feed water contaminants at 5-20 times higher concentration, it can usually be recycled back to the RO pre-treatment. The recycle option increases the total plant recovery and so minimizing the valuable total water consumption.
Summary of EDI features:
- Environmental friendly, no regeneration chemicals are needed.
- EDI is a continuous process and produces a constant quality.
- EDI systems are extremely compact and require minimum footprint.
- Low operating cost, electricity only.
- Very fair capital cost.
- Minimum facility requirements and operator attention.
- High system recovery.
- 1000’s of systems installed worldwide up to a capacity of 1.500 m3/h.


Beneficial EDI characteristics
The electrodeionization technology distinguishes itself as a continuous ion exchange technique for the production of (ultra) pure water without the need for chemical regeneration and wastes neutralization steps. During the process, the polishing part of the resin is continuously regenerated. In comparison with the more traditional ion exchange systems, this reduces facility costs such as waste neutralization equipment and hazardous fumes ventilation. The lack of chemical usage, handling and storage help to improve health and safety, as well as corrosion prevention in facilities and equipment. EDI modules can be fed with feed water conductivity equivalent up to 40 µS/cm and water hardness up to 1 ppm, allowing a wider range of applications. Electro deionization systems can achieve up to 99,9 % salt removal, reduce the levels of individual ionic species to parts-per-billion or even parts-per-trillion levels, and produce high-purity water with a resistivity of 10 to 18,2 MegOhm.cm (0,1 to 0,055 microS/cm conductivity). Since the EDI concentrate (or reject) stream contains the feed water contaminants at 5-20 times higher concentration, it can usually be recycled back to the RO pre-treatment. The recycle option increases the total plant recovery and so minimizing the valuable total water consumption.
Summary of EDI features:
- Environmental friendly, no regeneration chemicals are needed.
- EDI is a continuous process and produces a constant quality.
- EDI systems are extremely compact and require minimum footprint.
- Low operating cost, electricity only.
- Very fair capital cost.
- Minimum facility requirements and operator attention.
- High system recovery.
- 1000’s of systems installed worldwide up to a capacity of 1.500 m3/h.
Contact us for a tailor-made solution
To provide you with the right EDI water treatment system for your business, we will first need to be informed about your current situation. Please contact Deionx and tell us your needs. In return, we can offer you a tailor-made solution using either Ionpure MX, VNX, LX, or one of our other systems. Contact us by calling +31 164 265921.